英文総説・書籍章
Direct Reprogramming of Microglia into Neurons
Kanae Matsuda-Ito, Eri Hatai, Tomohiko Irie, Taito Matsuda, Kinichi Nakashima
Methods in Molecular Biology, vol. 2974, Lentiviral Vectors. Humana Press, New York, NY (2026).
https://doi.org/10.1007/978-1-0716-4807-0_13
|
Tripartite Interaction of Epigenetic Regulation, Brain Aging, and Neuroinflammation: Mechanistic Insights and Therapeutic Implications |
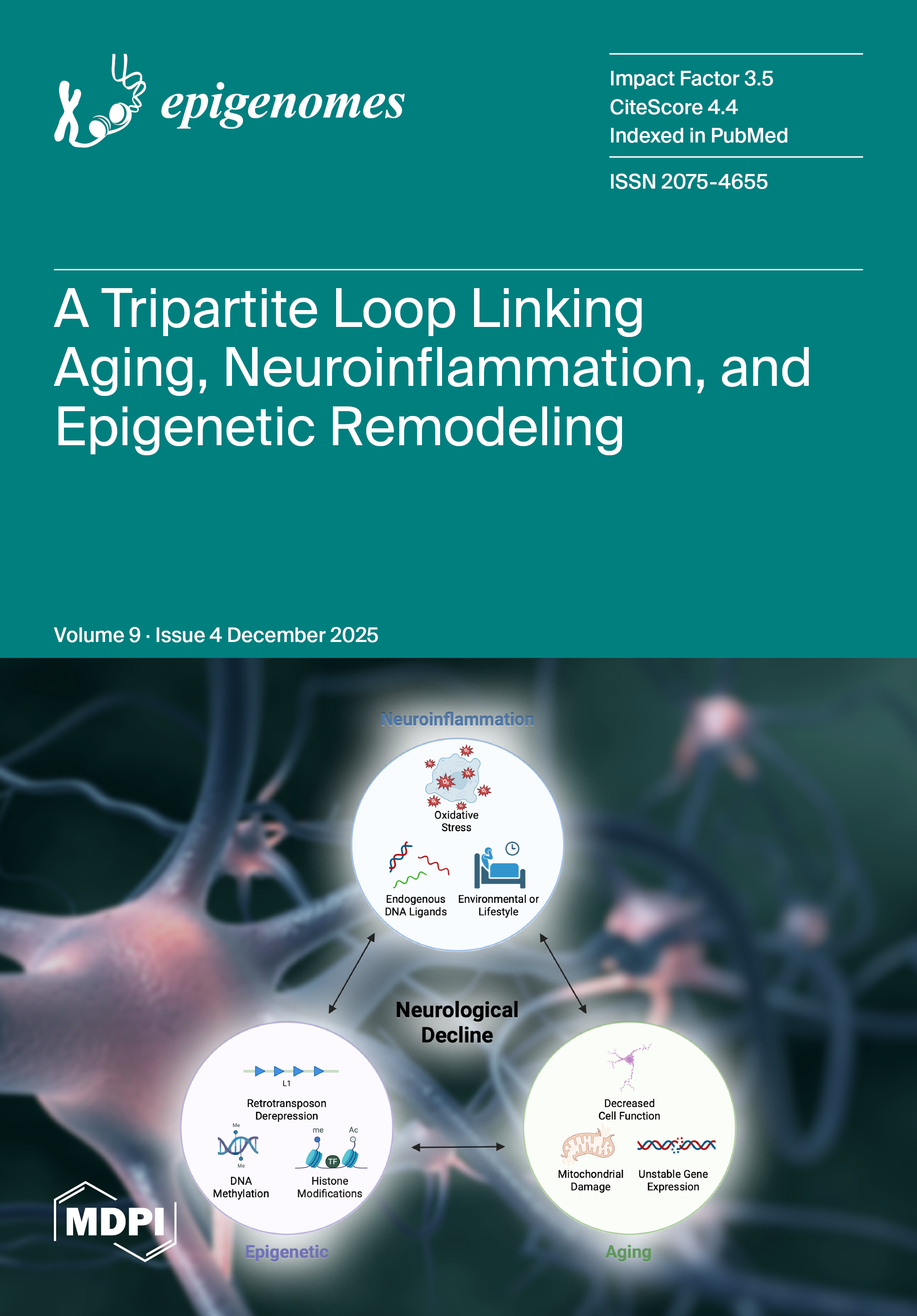
|
The influence of the choroid plexus on brain function: beyond its role in cerebrospinal fluid production
Sayako Katada, Kelren S. Rodrigues, Kinichi Nakashima
Inflammation and Regeneration, 45, Article number: 20 (2025).
https://doi.org/10.1186/s41232-025-00386-1
Epigenetic Regulation of Neural Stem Cells in Developmental and Adult Stages
Shu Kunoh, Hideyuki Nakashima, Kinichi Nakashima
Epigenomes, 8(2) (2024).
https://doi.org/10.3390/epigenomes8020022
Lineage Reprogramming: Genetic, Chemical, and Physical Cues for Cell Fate Conversion with a Focus on Neuronal Direct Reprogramming and Pluripotency Reprogramming
Taichi Umeyama, Taito Matsuda, Kinichi Nakashima
Cells, 13(8) (2024).
https://doi.org/10.3390/cells13080707
Multifaceted Regulation of Neural Stem Cell Fate in the Developing Brain
Takanobu Uotsu, Kinichi Nakashima, Sayako Katada
Medical Research Archives, 12(3) (2024).
https://doi.org/10.18103/mra.v12i3
Epigenetic memory of drug exposure history controls neural stem cell quiescence in the adult brain
Masakazu Iwamoto, Taito Matsuda
Neural Regen Res, 4 (2024).
https://pubmed.ncbi.nlm.nih.gov/37843197/
Seizure-induced hilar ectopic granule cells in the adult dentate gyrus
Yuka Kasahara, Hideyuki Nakashima, Kinichi Nakashima
Front Neurosci, 17, 1150283 (2023).
https://pubmed.ncbi.nlm.nih.gov/36937666/
Regulation of Adult Mammalian Neural Stem Cells and Neurogenesis by Cell Extrinsic and Intrinsic Factors
Shuzo Matsubara, Taito Matsuda*, Kinichi Nakashima*
Cells, 10, 1145 (2021).
https://doi.org/10.3390/cells10051145
In vivo direct reprogramming as a therapeutic strategy for brain and retina repair
Sekiryu H., Matsuda T.
Neural Regen Res, 10, 1998–1999 (2021).
https://pubmed.ncbi.nlm.nih.gov/33642376/
Natural and forced neurogenesis in the adult brain: Mechanisms and their possible application to treat neurological disorders
Matsuda T., Nakashima K.
Neurosci Res, 166, 1–11 (2021).
https://pubmed.ncbi.nlm.nih.gov/32497571/
Epigenetic Regulation for Acquiring Glial Identity by Neural Stem Cells during Cortical Development
Nakagawa T., Yoshikuni W., Sayako K., Yusuke K.
Glia, 68(8), 1554–1567 (2020).
New aspects of glioblastoma multiforme revealed by similarities between neural and glioblastoma stem cells
Kawamura Y., Takouda J., Yoshimoto K., Nakashima K.
Cell Biol Toxicol, Epub ahead of print (2018).
Epigenetic regulation of neural stem cell differentiation towards spinal cord regeneration
Kameda T., Imamura T., Nakashima K.
Cell Tissue Res, 371, 189–199 (2018).
Neural stem cell therapy aiming at better functional recovery after spinal cord injury
Zhu Y., Uezono N., Yasui T., Nakashima K.
Dev Dyn, 247, 75–84 (2018).
Emerging mechanisms underlying astrogenesis in the developing mammalian brain
Takouda J., Katada S., Nakashima K.
Proc Jpn Acad Ser B Phys Biol Sci, 93, 386–398 (2017).
Detection of bidirectional promoter-derived lncRNAs from small-scale samples using pre-amplification-free directional RNA-seq method
Hamazaki N., Nakashima K., Hayashi K., Imamura T.
Methods Mol Biol, 1605, 83–103 (2017).
Manipulation of promoter-associated noncoding RNA in mouse early embryos for controlling sequence-specific epigenetic status
Hamazaki N., Nakashima K., Imamura T.
Methods Mol Biol, 1543, 271–282 (2017).
Emerging roles for miRNA-based post-transcriptional regulation in neuronal morphogenesis and neurodevelopmental disorders
Tsujimura K., Nakashima H., Irie K., Nakashima K.
RNA Dis, 5, e1375 (2016).
Epigenetic regulation of neural stem cell property from embryo to adult
Murao N., Noguchi H., Nakashima K.
Neuroepigenetics, 3, 1–10 (2016).
Bidirectional communication between the innate immune and nervous systems for homeostatic neurogenesis in the adult hippocampus
Matsuda T., Nakashima K.
Neurogenesis, 2 (2015).
Epigenetic mechanisms regulating differentiation of neural stem/precursor cells
Adefuin A.M., Kimura A., Noguchi H., Nakashima K., Namihira M.
Epigenomics, 6, 637–649 (2014).
Epigenetic setting and reprogramming for neural cell fate determination and differentiation
Imamura T., Uesaka M., Nakashima K.
Philos Trans R Soc Lond B Biol Sci, 369, 1652 (2014).
Roles of Epigenetics in the Neural Stem Cell and Neuron
Yamamoto N., Uesaka M., Imamura T., Nakashima K.
Epigenetics in Psychiatry, 51–78 (2014).
Mechanisms of astrocytogenesis in the mammalian brain
Namihira M., Nakashima K.
Curr Opin Neurobiol, 23, 921–927 (2013).
Epigenetic regulation of neural stem cell fate during corticogenesis
MuhChyi C., Juliandi B., Matsuda T., Nakashima K.
Int J Dev Neurosci, 31, 424–433 (2013).
Epigenetic regulation in neural stem cell differentiation
Juliandi B., Abematsu M., Nakashima K.
Dev Growth Differ, 52, 493–504 (2010).
Epigenetic mechanisms in sequential differentiation of neural stem cells
Sanosaka T., Namihira M., Nakashima K.
Epigenetics, 4, 89–92 (2009).
Epigenetic mechanisms regulating fate specification of neural stem cells
Namihira M., Kohyama J., Abematsu M., Nakashima K.
Philos Trans R Soc Lond B Biol Sci, 363, 2099–2109 (2008).
Mechanisms of neural stem cell fate determination: extracellular cues and intracellular programs
Abematsu M., Smith I., Nakashima K.
Curr Stem Cell Res Ther, 1, 267–277 (2006).
Mechanisms underlying cytokine-mediated cell-fate regulation in the nervous system
Nakashima K., Taga T.
Mol Neurobiol, 25, 233–244 (2002).
